4 Dec 2017
Canine cardiology developments

Ultimate success in managing heart disease in dogs would be a cure. This is not yet possible, but the onset of congestive heart failure can be delayed by starting treatment with pimobendan during the preclinical stages of both degenerative mitral valve disease (DMVD) and dilated cardiomyopathy.
Biomarkers and continuous-loop ECG recorders offer non-invasive ways of monitoring cardiac disease and its progression, and offer an enhanced ability to detect intermittent arrhythmias, a potential cause of syncope. Cardiac troponins can provide an estimate of the severity of myocardial injury, and measurement of the N-terminal pro-brain natriuretic peptide can help differentiate cardiac from non-cardiac causes of respiratory distress.
Improved heart rate control, with a combination of diltiazem and digoxin in patients with atrial fibrillation, and the availability of the potent diuretic torasemide – which can be used in cases of furosemide resistance – improve veterinarians’ ability to medically manage refractory and potentially fatal complications of heart disease in dogs.
Cardiac surgery, specifically mitral valve repair, has shown promise in managing DMVD in dogs and may become the future treatment of choice, as in humans.
The available genetic tests for heart disease (cardiomyopathies) discussed in this article do not offer UK breeders the confidence to identify carriers or at-risk dogs.
The outcome of the “Evaluating pimobendan in dogs with cardiomegaly: the EPIC study” (Boswood et al, 2016) is anticipated to have an enormous impact on the management of degenerative mitral valve disease (DMVD).
The Boehringer Ingelheim-funded study investigated the possibility of extending the preclinical period of DMVD by pharmacological intervention. Specifically, it determined whether a dog with stage B2 DMVD (Atkins et al, 2009) – a heart murmur higher than or equal to grade three, cardiomegaly (dilated left ventricle and/or enlarged left atrium on echocardiography, or a vertebral heart score more than 10.5 on a lateral thoracic radiograph) – would benefit from pimobendan dosed between 0.4mg/kg/day and 0.6mg/kg/day (Figures 1 to 3).
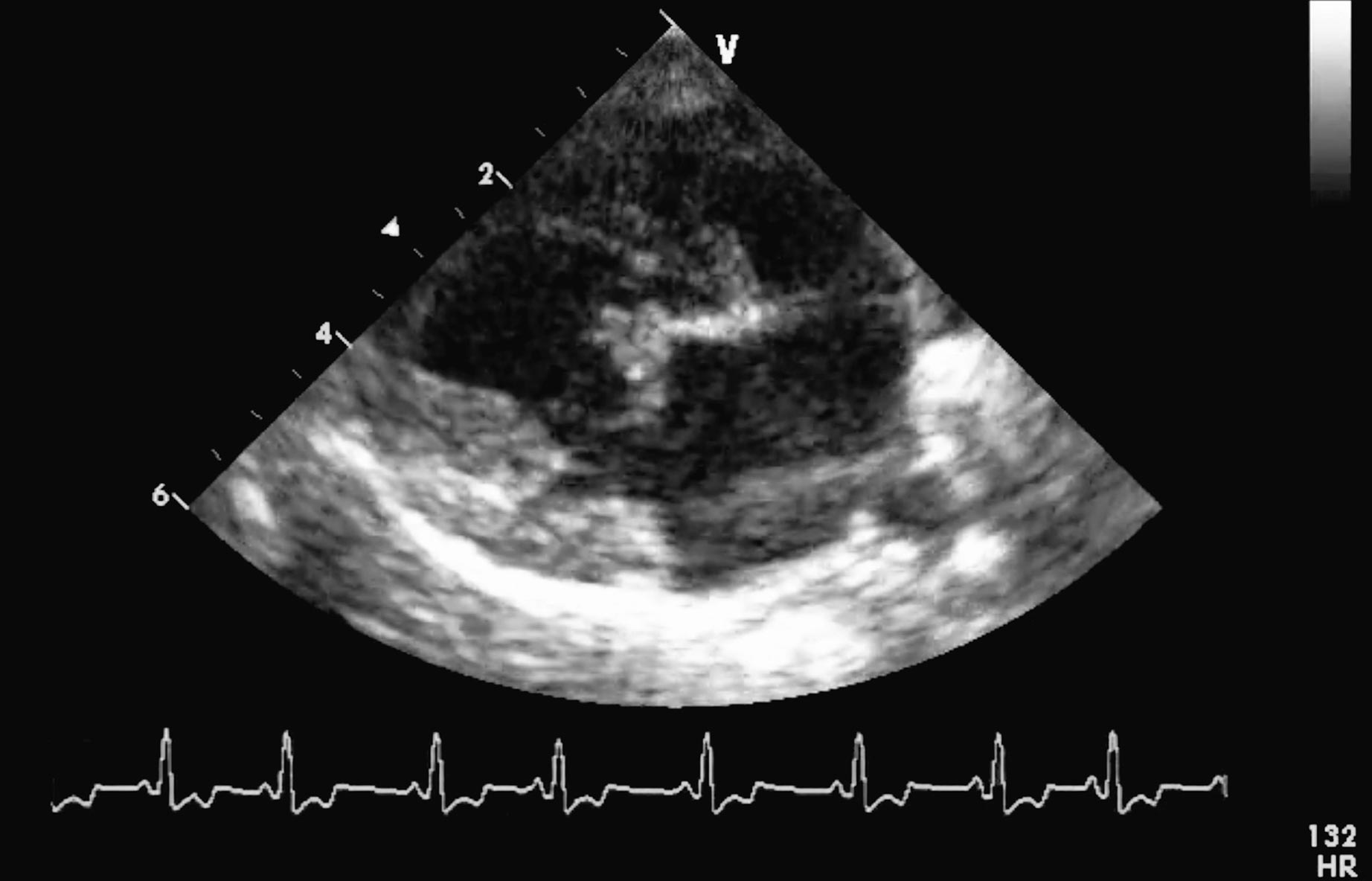
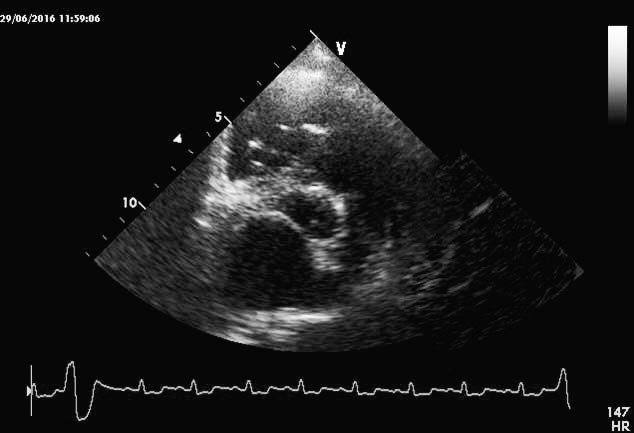
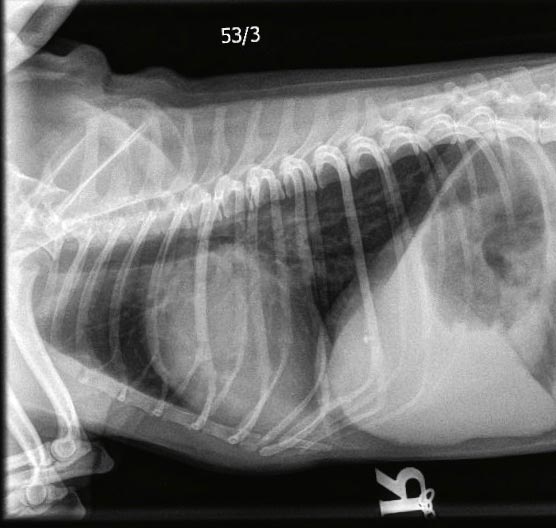
The number of dogs reaching the primary end point of the study – death from cardiac disease (spontaneous or euthanasia) or congestive heart failure – was similar in the pimobendan-treated and placebo-treated groups. However, the major benefit – and, therefore, the clinical relevance of this study – was the time to reach this end point was significantly longer in dogs treated with pimobendan (1,228 days, compared to 766 days). This translates to about 15 months of additional good quality of life and a reduction of risk of developing congestive heart failure by a third. Boehringer Ingelheim has obtained this indication in its licence for pimobendan.
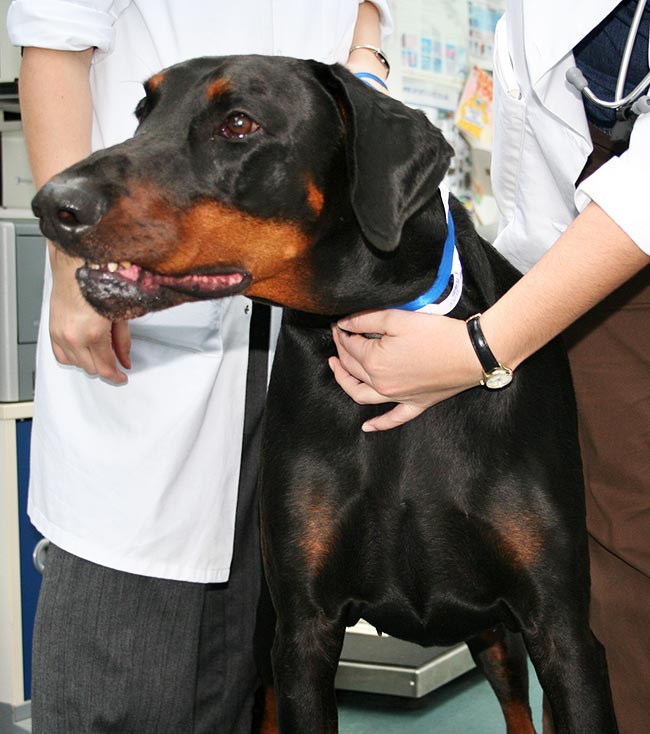
In addition, the “Efficacy of pimobendan in the prevention of congestive heart failure or sudden death in Dobermann pinschers with preclinical dilated cardiomyopathy (the PROTECT study)”, by Summerfield et al (2012), also Boehringer Ingelheim-funded, has had a major impact on the way the occult (preclinical) stage of dilated cardiomyopathy (DCM) is managed.
Pimobendan administered to Dobermanns with occult DCM significantly delayed the onset of congestive heart failure or occurrence of sudden death, compared to Dobermanns receiving the placebo.
Despite a protracted preclinical phase – more than two to three years – of DCM in this breed, survival time is limited to an average of two to four months once congestive heart failure has developed, with a 10% survival reported one year from diagnosis.
The survival time was longer in Dobermanns receiving pimobendan (718 days) than those not receiving the drug (441 days). This translated to a nine-month longer preclinical stage, which significantly improved the quality of life of patients.
Although this study specifically examined the benefit of pimobendan in Dobermanns (Figure 4) with occult DCM, these findings are generally extrapolated to other breeds with occult DCM due to the likely survival benefit.
Biomarkers in canine cardiology
Two commercially available, cardiac-specific biomarkers are commonly employed – plasma N-terminal pro-brain natriuretic peptide (NT-proBNP) and cardiac troponin I (cTnI), including the high-sensitivity cardiac troponin assay. However, their value in diagnosing and monitoring heart disease in dogs is confounded by a number of patient co-morbid diseases, including renal disease, and – specifically with NT-proBNP – poor repeatability and high variance, to the extent breed and disease stage reference ranges are being proposed (Winter et al, 2017).
The main indication given by the manufacturer for using NT-proBNP is as a cost-effective test to support potentially costly referral, and monitoring of dogs after detection of a heart murmur on auscultation. NT-proBNP more than 1,500pmol/L has been found to be a strong risk factor for the development of congestive heart failure in dogs with DMVD (Reynolds et al, 2012).
The second generation NT-proBNP ELISA assay has an increased upper limit and, like the first generation assay, is able to discriminate cardiac disease severity non-invasively, which is helpful in evaluating our at-risk population – geriatric, small breed dogs with murmurs (Chetboul et al, 2009; Serres et al, 2009; DeFrancesco et al, 2007; Fox et al, 2015).
NT-proBNP is also an ideal marker for distinguishing the cause of respiratory distress, with a cut-off of 2,447pmol/L being a reasonably sensitive way of distinguishing congestive heart failure from non-cardiogenic respiratory distress or, more specifically, excluding congestive heart failure (Fox et al, 2015; DeFrancesco et al, 2007).
NT-proBNP has also been shown to be increased in dogs with experimental aortic stenosis and could be used as a screening test, together with clinical signs in young at-risk dogs (Hori et al, 2008).
Cardiac troponins are markers of myocardial injury and have the advantage of species homology, allowing veterinarians to use human laboratories for testing. Their concentrations are proportional to the degree of injury, and the very short half-life – 1.86 hours for cTnI – allows monitoring of progression or recovery from the cardiac insult (Langhorn and Willesen, 2016).
The disadvantage of this biomarker is specificity as to the aetiology of the injury is lacking and many types of assay are reported in the literature, making comparisons between studies difficult.
Myocardial injury is described in dogs with non-cardiac disease, including sepsis, toxicities and envenomation, hypotension, hypoxia, anaemia, thrombotic conditions, reperfusion injury with resuscitation, and cardiac-related injuries, including epicardial or endocardial haemorrhage, cardiotoxic (doxorubicin) and vasopressor drugs, myocardial wall stretch, arrhythmias and tachycardia (Langhorn and Willesen, 2016).
In some instances, troponin values may exceed 10ng/ml, reference less than 0.11ng/ml. Clinically, they are helpful in their negative predictive value due to their sensitivity – lack of an increase above reference can exclude myocardial injury – and being considered a prognosticator in various cardiac injuries, in addition to offering insight into the nature of the cardiac injury in terms of whether it is reversible or whether the underlying insult has resolved.
One should be mindful troponins are only modestly raised in dogs with primary cardiac disease (less than 0.3ng/ml), but concentrations can be predictive of outcome in the case of canine cardiomyopathies – with cTnI more than 0.2ng/ml indicating shorter survival (112 days), in contrast with dogs with cTnI less than 0.2ng/ml (357 days). Concentrations have also been found to correlate with cardiac remodelling, specifically left atrial and ventricular size (Oyama and Sisson, 2004). Ideally, troponin should be interpreted together with a marker of myocardial stretch, such as NT-proBNP (Hezzell et al, 2012).
ECG loop recorders and alternative devices for syncopal dogs
Syncope is a common complaint in veterinary medicine and it remains critical to rule out potentially life-threatening conditions while diagnosing conditions that may benefit from therapy (Cheung and Krahn, 2016).
Implantable loop recorders (ILR) are lead-free SC devices that continuously monitor the heart rhythm and record events over a time frame, measured in years (Tomson and Passman, 2015). They have become an important pillar in the diagnostic work-up of human patients with unexplained syncope (Cheung and Krahn, 2016) and are available for veterinary use. Remote monitoring capabilities and miniaturisation have improved the acceptability of this diagnostic strategy, and studies in humans have advocated for an earlier implantation of ILRs to reduce costs of syncope assessment (Cheung and Krahn, 2016).
A summation of randomised trials for patients with unexplained syncope pointed towards a benefit of prolonged monitoring with an ILR over conventional investigations (Cheung and Krahn, 2016). In human patients with unexplained syncope, standard Holter monitoring was found to be non-diagnostic in up to 90% of patients (Linzer et al, 1990). It has also been suggested the ILR may play a role in the assessment of patients with uncertain diagnoses of epilepsy (Petkar et al, 2012).
Given the wide availability of smartphones, the diagnostic accuracy of a smartphone electrocardiograph was assessed for evaluating heart rhythm and ECG measurement in dogs (Vezzosi et al, 2016). This device was found reliable for evaluation of heart rate, rhythm, atrioventricular blocks and ectopic beats in a study of 166 dogs. The analysis of smartphone tracings showed a 100% sensitivity and 97.9% specificity in detecting arrhythmias, and was thought to represent an additional tool in the management of dogs with arrhythmias (Vezzosi et al, 2016).
Rate control in atrial fibrillation
In atrial fibrillation (AF), heart rate control is aimed at slowing the ventricular response rate to rapid impulses from the atria, by increasing the filtering function of the atrioventricular (AV) node (Pariaut, 2017).
Most cases of AF are diagnosed in dogs with structural heart disease, often presenting in congestive heart failure (Menaut et al, 2005). Not all animals in AF require treatment, and based on the ventricular response rate, clinical signs and structural cardiac changes, a decision on whether to initiate treatment can be made (Pariaut, 2017). Maximum mean heart rate above which treatment should be initiated, and the target heart rate that should be achieved, by a rate control strategy are not well defined. A 24-hour mean heart rate of 140bpm or less has been proposed as adequate for dogs with AF (Pariaut, 2017).
Prolonging the refractory period of the calcium-dependent nodal cells is a preferred rate-control approach to increase AV node filtering (Pariaut, 2017). Diltiazem can be administered as an IV bolus and constant-rate infusion (CRI) for acute management of tachyarrhythmias, although the IV formulation of this drug is not readily available in the UK.
Esmolol, a short-acting beta-blocker, can be administered as a repeated bolus or CRI. The negative inotropic effect of this drug needs to be considered as a potential contraindication in a dog with heart failure. For long-term rate control, oral diltiazem is widely used for management of AF. Diltiazem monotherapy is rarely effective enough, however, and better rate control can be achieved by adding digoxin to slow AV conduction by increasing vagal tone (Pariaut, 2017).
Restoration of sinus rhythm by electrical cardioversion was retrospectively studied in a small group of dogs with AF and heart disease. The results suggested AF reoccurred in 50% of dogs within approximately two months – and in most dogs by five months – after cardioversion (Bright and ZumBrunnen, 2008; Bright et al, 2005).
High vagal tone is recognised as a major trigger for initiating AF by promoting re-entrant arrhythmias (Pariaut et al, 2008). Pharmacological cardioversion with lidocaine or electrical cardioversion can be tried, if necessary, in these acute cases. It is important to treat systemic conditions that can serve as a trigger for this arrhythmia – for example, endocrine disease (Pariaut, 2017). Treatment options for AF are summarised in Table 1.
| Table 1. Pharmacological treatment of dogs with AF (modified from Pariaut, 2017) | ||
|---|---|---|
| Drug | Treatment strategy | Dosage |
| Lidocaine (Class 1b; sodium channel blocker) | Rhythm control, vagally mediated AF | Slow IV bolus: 2mg/kg (maximum four boluses) |
| Atenolol (Class 2; beta-blocker) | Rate control | 0.2mg/kg to 1mg/kg q12-24h PO |
| Esmolol (Class 2; beta-blocker) | Rate control | Slow IV bolus: 0.2mg/kg to 0.5mg/kg CRI: 0.05mg/kg/min to 0.25mg/kg/min |
| Sotalol (mixed Class 2 and 3; potassium channel blocker) | Mild rate control, ventricular tachyarrhythmias during AF | 1mg/kg to 3mg/kg q12h PO |
| Amiodarone (Class 3; potassium channel blocker) | Rhythm control, mild rate control | Loading dose: 10mg/kg to 30mg/kg q24h for 2-14d PO Maintenance dose: 6mg/kg to 15mg/kg q24h PO |
| Diltiazem (Class 4; calcium channel blocker) | Rate control | IV bolus: 0.1mg/kg to 0.9mg/kg over 5 min CRI: • 0.05mg/kg/h to 0.15mg/kg/h • 1mg/kg to 2mg/kg q8h PO • 3mg/kg to 5mg/kg q12h PO (long-acting) |
| Digoxin (cardiac glycoside) | Mild rate control | 0.005mg/kg q12h PO |
| AF – atrial fibrillation; CRI – constant-rate infusion. | ||
Congestive heart failure: new developments in diuretic therapy
Loop diuretics, such as furosemide and torasemide, are important in the treatment of congestive heart failure (Cody et al, 1994).
Torasemide acts on the thick ascending limb of Henle’s loop to promote rapid and marked excretion of water, sodium and chloride (Cody et al, 1994). Torasemide has been demonstrated to have more predictable pharmacokinetics and pharmacodynamics, and a lower influence on electrolyte disorders, so has improved tolerability compared to furosemide (Kido and Ohtaki, 2001). Furthermore, torasemide has been shown to inhibit aldosterone secretion in vitro (Goodfriend et al, 1998) and reduce myocardial fibrosis in chronic human heart failure patients (Cosín and Díez, 2002).
It has been shown torasemide is an efficacious and potent diuretic drug in the dog (Paulin et al, 2016). The typical dosage of 0.1mg/kg to 0.3mg/kg by mouth every 24 hours has been shown to be 10 times more potent than furosemide and results in less development of diuretic tolerance than furosemide (Côté, 2017).
Valve repair
Surgical therapy for mitral valve disease, including valve replacement and valve repair, is considered the standard of care in humans.
With cardiopulmonary bypass more recently available for small breeds, open-heart surgery is now a feasible option for managing DMVD and mitral valve dysplasia. Abnormal mitral valves can be replaced by mechanical valves or bioprosthetic valves. The latter are ideal for dysplastic valves, as they are so abnormal in these conditions they cannot be repaired.
Valve replacement requires careful matching of the valve to the patient annulus size and ongoing need for antithrombotic therapy. In humans, a long-term complication risk is valve degeneration and calcification, but this has not been reported in dogs, with survival of more than 17 months in the few cases described.
Open-heart surgery – with cardiopulmonary bypass, circumferential annuloplasty and artificial chordae tendineae replacement – is considered the surgical therapy of choice for DMVD, with improved clinical outcome, more than three years’ survival in 93% of cases and no need for ongoing antithrombotic therapy (Mizuno et al, 2013; Uechi, 2012; Uechi et al, 2012).
Tricuspid valve stenosis has been treated with per catheter valvuloplasty techniques in a limited number of dogs, but although most dogs clinically improved, many had recurrence of clinical signs (Lake-Bakaar et al, 2017). Similarly, tricuspid valve replacement with porcine or bovine valves has been reported to be associated with a high incidence of complication (Bristow et al, 2017).
Genetic tests for Dobermann and boxer cardiomyopathies
Genetic abnormalities have been linked to the development of many cardiac conditions in humans and with hypertrophic cardiomyopathy in Maine Coon and Ragdoll cats. However, the identification of genetic causes of the major heart diseases in dogs has been less successful, despite being a major area for research.
DCM is common in Dobermanns (Figure 5) and associated with high mortality. A US study found a mutation in a gene encoding a mitochondrial protein, which was associated with the development of DCM in Dobermanns, and a genetic test based on this is available (Meurs et al, 2012). However, as no association has been found between this mutation and DCM in Dobermanns in Europe, this test is not likely to be of value in practice in the UK (Owczarek-Lipska et al, 2013).

Figure 5. A Dobermann being fitted with a 24-hour Holter ECG monitor to determine the severity of ventricular ectopy.
The most common adult-onset cardiac disease in boxers (Figure 6) is arrhythmogenic right ventricular cardiomyopathy (ARVC), which is strongly familial in boxers. Several genetic mutations associated with ARVC have also been found in humans. In 2010, a novel mutation of the gene encoding for striatin was found and appeared to be a convincing candidate for the cause of ARVC in boxers (Meurs et al, 2010). A genetic test based on this was available, but subsequent use in the US and UK has shown no correlation between having the mutation and developing ARVC. A subsequent extensive analysis of pedigrees of UK boxers has shown the striatin mutation is not causal, but may be close to the (as yet unknown) causal mutation for ARVC in boxers on the same chromosome (Cattanach et al, 2015) and may be related to severity. However, the striatin test seems to be of no clinical value in UK practice.

Conclusion
Improving and extending quality of life for patients are factors most valued by clients, as the negative impact of chronic management of congestive heart failure includes the impact on client work and social life, constant need for veterinary visits, worry the dog may need hospitalisation, the difficulty of leaving it in a boarding facility and concern it may be suffering.
With several new therapeutic interventions and screening tests in preclinical stages of heart disease, we can now achieve this. In addition, more efficacious drugs and dosing protocols exist for treating congestive heart failure and arrhythmias.
Finally, valve repair will offer exciting curative options for the most commonly acquired heart diseases in veterinary practice.
- Some drugs mentioned are used under the cascade.
