10 Oct 2023
Growing mycobacterial infections in cats and dogs: the challenges
Conor O’Halloran, Danièlle Gunn-moore and Petra Černá discuss the features of an increasingly encountered clinical condition in companion animal practice and the treatment options available.

Image © julijadmi / Adobe Stock
Mycobacterial infections are an increasingly encountered clinical condition in companion animal practice but they remain challenging to manage for a number of reasons.
Firstly, they can be difficult to diagnose definitively and it can be even harder to identify the infecting species. Beyond this, their treatment is prolonged and patient compliance can be challenging, as well as raising potential zoonotic transmission risks.
The aim of this article is to briefly cover the main species of mycobacteria that infect cats and dogs in the UK as well as their clinical signs, diagnostic testing options and treatment considerations.
Causes of infection
Mycobacteria comprise a genus of highly related bacterial species that share a characteristic thick, hydrophobic and mycolic acid-rich cell wall, which is predominately made of peptidoglycan and arabinogalactan and confers the typical acid-fast staining pattern used to aid in their diagnosis1-4.
Many species of mycobacteria have been identified, in particular since the instigation of phylogenetic analyses, which is capable of discriminating different species that would not have previously been possible (such as those that are not possible to culture in vitro)1,4. This has now largely replaced the previous Runyon system, which determined species identity based on laboratory growth characteristics including time to form colonies, substrate usage and colony pigmentation1,4.
However, the most useful classification system to refer to clinically is based on pathogenicity and divides the obligately pathogenic Mycobacterium tuberculosis complex (MTBC) from the non-tuberculous mycobacteria.
As the nomenclature implies, the MTBC organisms all cause clinical tuberculosis (TB) in mammalian species, including companion animals.
In cats, TB can be caused by either M bovis or M microti, whereas dogs can additionally be infected with M tuberculosis1-4. The reason for feline resistance to M tuberculosis infection is not understood, but previous reports of this organism being isolated from cat tissue samples predate the use of molecular methods and robust sub-classification of the group, and so are now thought to have been erroneous. This is significant as any animal infected with M tuberculosis should be euthanised and the appropriate health agencies (for example, the Animal and Plant Health Agency and the Health Security Agency) notified due to the public health risks.
All of the other mycobacteria are, therefore, collectively referred to as the non-tuberculous mycobacteria (NTM), although they are occasionally named as “atypical” mycobacteria or “mycobacteria other than tuberculosis” (or MOTT) by some authors.
With respect to companion animals, some groups, terms and complexes, of species cause very similar and overlapping clinical signs, but they can have different disease progression and optimal treatment strategies1-4. These include the M avium complex (MAC), which causes clinical signs that are highly convergent with TB, but which is, somewhat counterintuitively to most owners, significantly harder to treat (see later). The species within the MAC group are all slow growing mycobacteria (based on the Runyon classification), but others such as M malmoense have been isolated from feline tissue samples.
In contrast, rapidly growing mycobacteria exist, such as M smegmatis, as well as those that cannot be cultured under any known laboratory conditions, such as M lepraemurium, one of the causative agents of feline leprosy5.
Only one study has quantified the relative incidence of each species of mycobacteria in cats, and none are available for dogs, due to the significantly lower prevalence of disease in this species generally1. The available data are based on culture results (as they predate commercially available PCR testing) and identified M microti most frequently (19% of samples), followed by M bovis (15%), M avium (7%), NTM other than M avium (6%) and no growth in 53% of cases1.
For all mycobacterial infections, the putative route of infection is the same. It is thought that nearly all companion animals most likely become infected from bites or wounds from smaller (prey) animals such as mice, voles or birds1-4. Once inoculated into the skin, the immune response gives rise to the classic granulomatous (or pyogranulomatous) inflammation at the area, usually in the skin, as well as reactive lymphadenomegaly (see later)1-4. The exceptions to this are where there is a history of raw meat feeding or potential access to other raw contaminated products, such as milk from severely affected cattle. In these cases, systemic signs are more prevalent and the clinical disease is generally more severe6.
Clinical signs
As has been alluded to, many of the mycobacteria, irrespective of their taxonomy, can cause very similar presenting clinical signs.
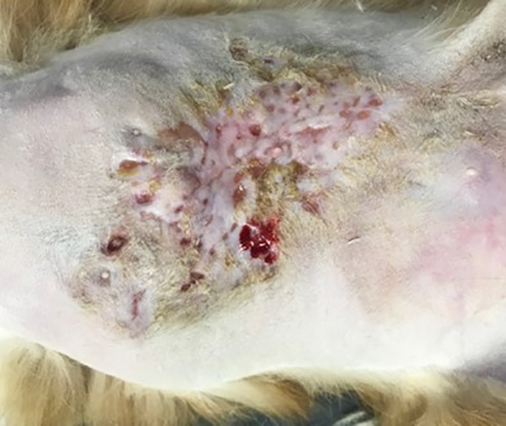
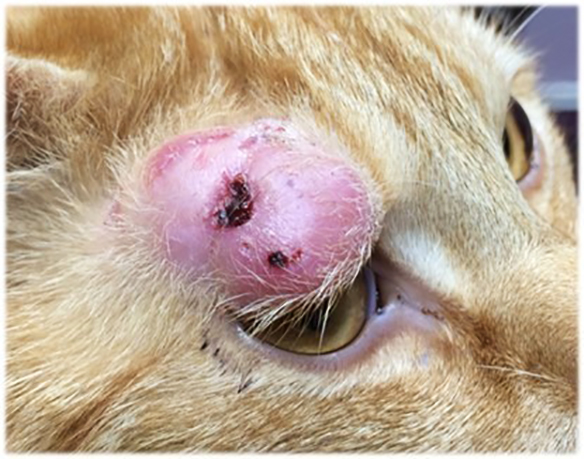
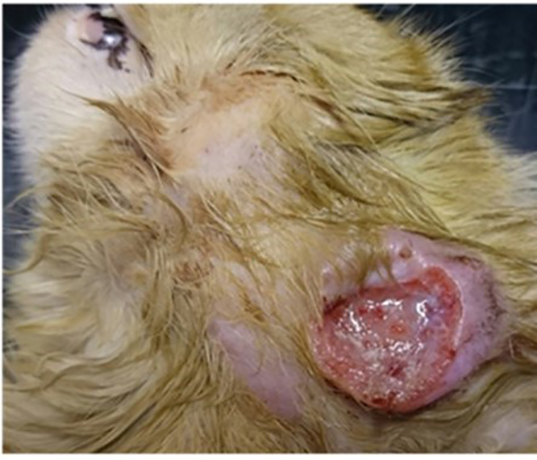
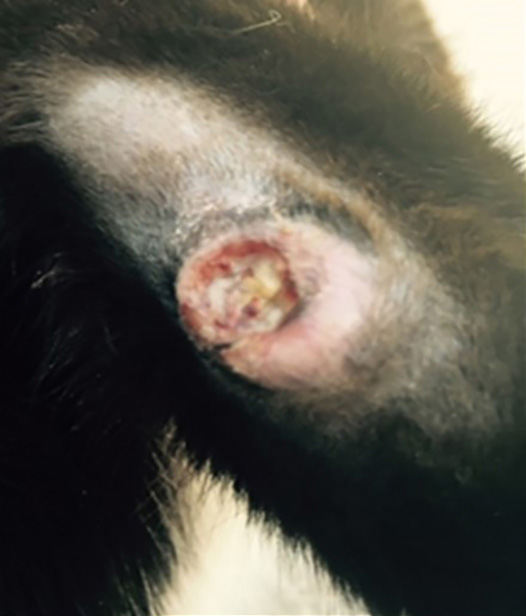
In cats, by far the most common presenting complaint is the presence of single or multiple cutaneous lesions, which are present in almost three-quarters of all cases1-4,7,8. These lesions are typically slow to develop and well demarcated and typically non-painful on palpation, although they are sometimes ulcerated or have discharging tracts that can be uncomfortable1-4. These lesions are located most frequently on the head or at the base of the tail.
As would be expected, the local lymph nodes (such as the submandibular nodes in cases of head lesions) are usually enlarged, but in some cases cats present with a generalised lymphadenopathy and these lymph node changes can also occur on their own, in the absence of the “primary” skin lesion1,4.
A number of clinical signs caused by mycobacteria have been noted with less frequency, including gastrointestinal manifestations, ocular changes (for example, chorioretinitis), joint involvement, spinal granulomas resulting in neurological deficits as well as primary pulmonary or generalised systemic disease1-4,7-9.
Previously, it was suggested that systemic or pulmonary signs were rare and only seen in approximately 10% of cases. However, in cases identified where raw food was the most likely source of infection, a higher proportion developed systemic illness manifesting as general lethargy and weight loss (32% of cases) and respiratory signs (72% of cases)6.
Dogs are less frequently affected by mycobacterial disease than cats and so there is less data analysing the clinical presentations. In general, however, dogs are diagnosed later in the course of disease, so multi-systemic illness is common by the time of diagnosis, though respiratory and gastrointestinal forms are also seen.
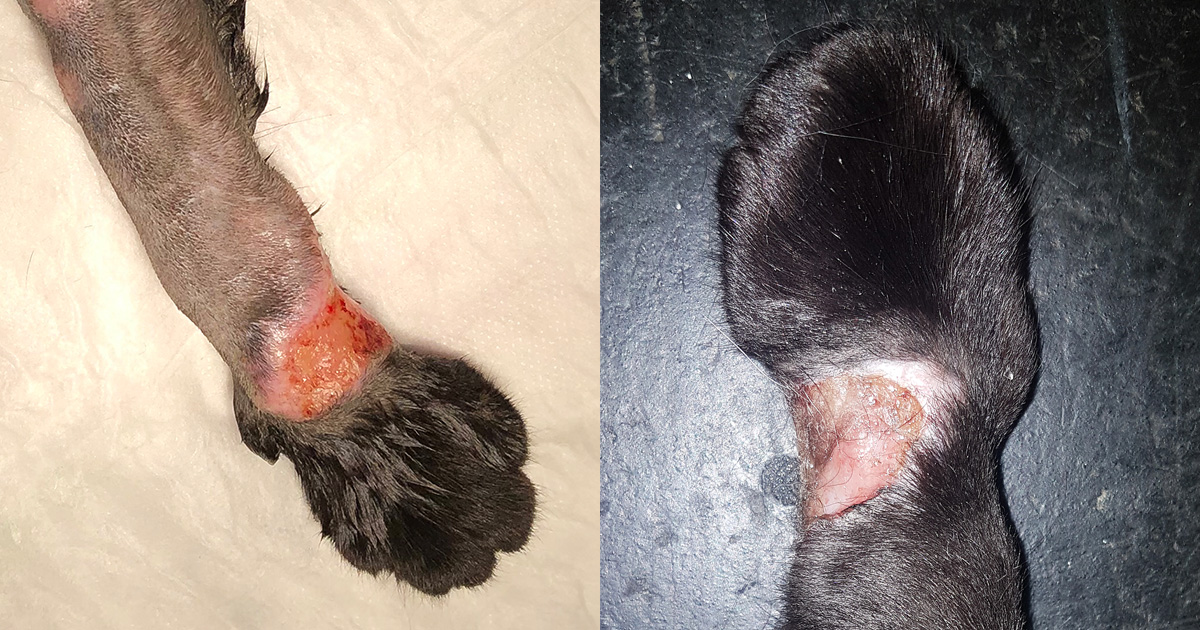
Diagnosis
The diagnosis of a mycobacterial infection and identification of the causative species remains challenging, in most part because the number of organisms (and, therefore, the sensitivity of some, but not all) diagnostic tests is variable, depending on the species, the duration of disease and the host immune response.
Given that a large majority of cases present with cutaneous nodules and/or lymphadenomegaly then cytology can be a useful initial diagnostic step, as fine needle aspiration is non-invasive and analysis can provide evidence against other differential diagnoses, such as lymphoma8. However, the presence of macrophages and neutrophils is often non-specific and, due to the small sample size acquired, the use of special stains, such as the commonly used Ziehl–Neelsen stain, can frequently fail to positively identify mycobacteria1,2.
Surgical removal and histopathology is often the single most useful diagnostic test for confirming mycobacterial infection, as the larger sample size provides the opportunity to identify the structure of the granuloma and for selection of the most likely sections to stain to maximise sensitivity4. When any skin nodule is removed where mycobacterial infection is considered possible then the tissue should ideally be halved, with one part submitted for histology and special stains and the other retained in a sterile sample pot in the freezer in case further testing, such as PCR or culture, is required.
If histology is diagnostic or highly suggestive of mycobacterial infection, then follow up investigations are indicated and focus should be on establishing the extent of the clinical disease in the patient and identifying the causative species to optimise treatment and maximise the chances of successful outcome – as well as to provide information to the owners about the zoonotic potential (or otherwise).
Patient testing
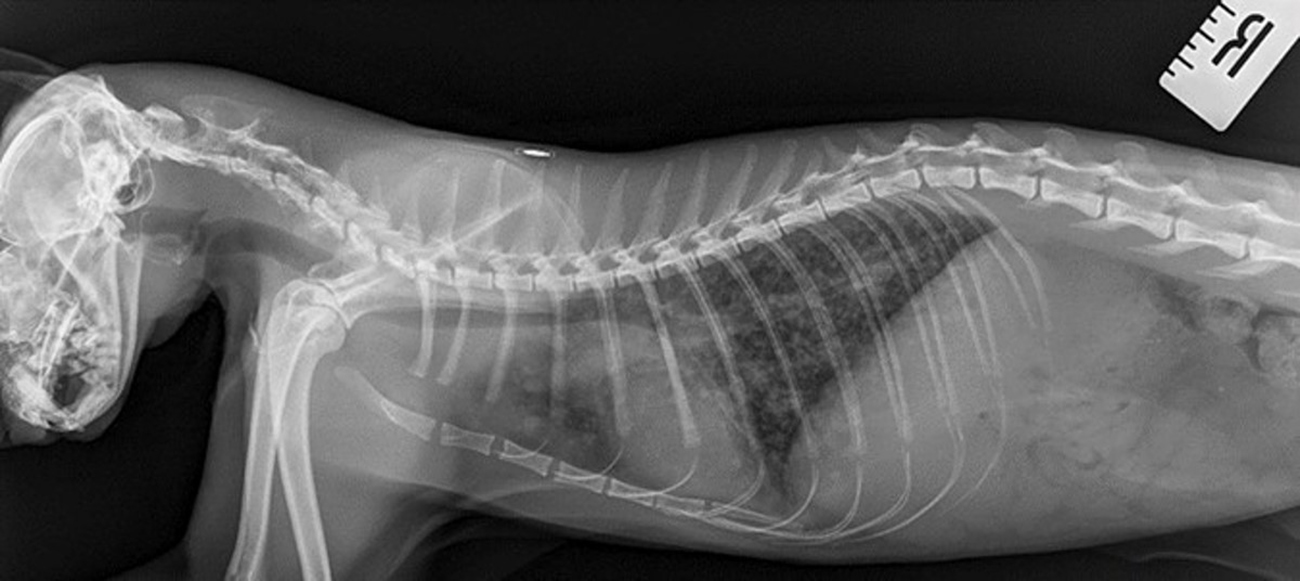
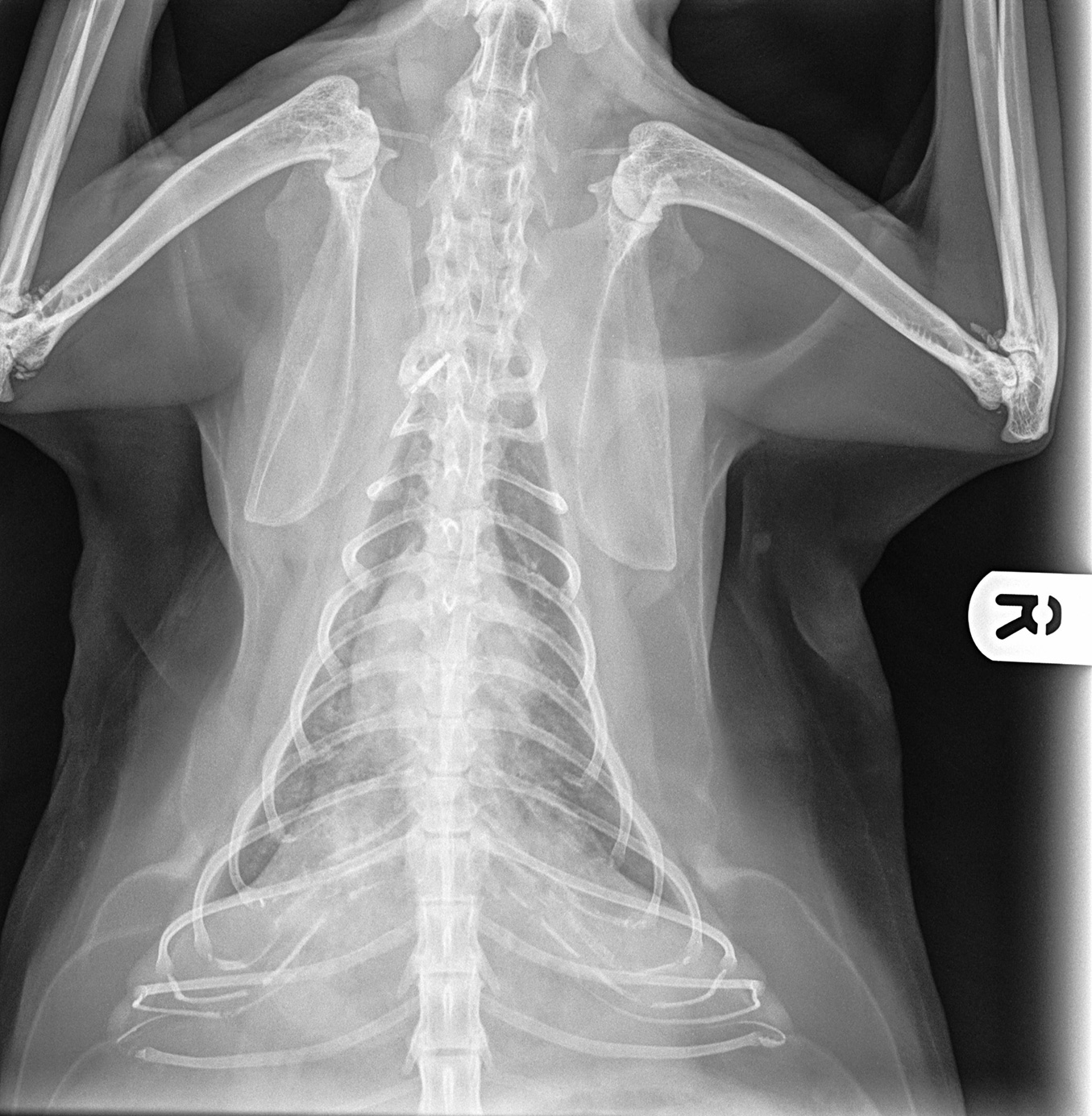
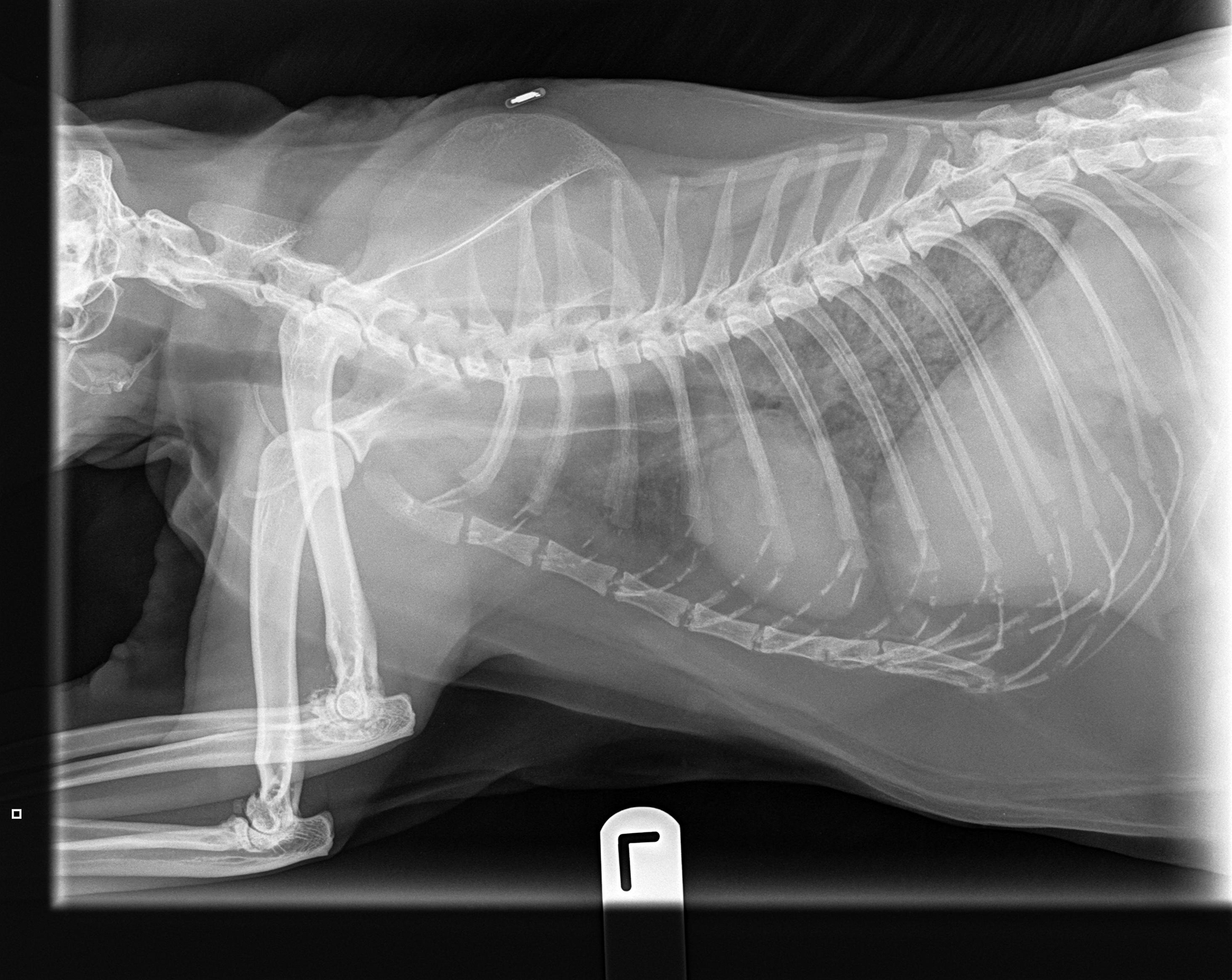
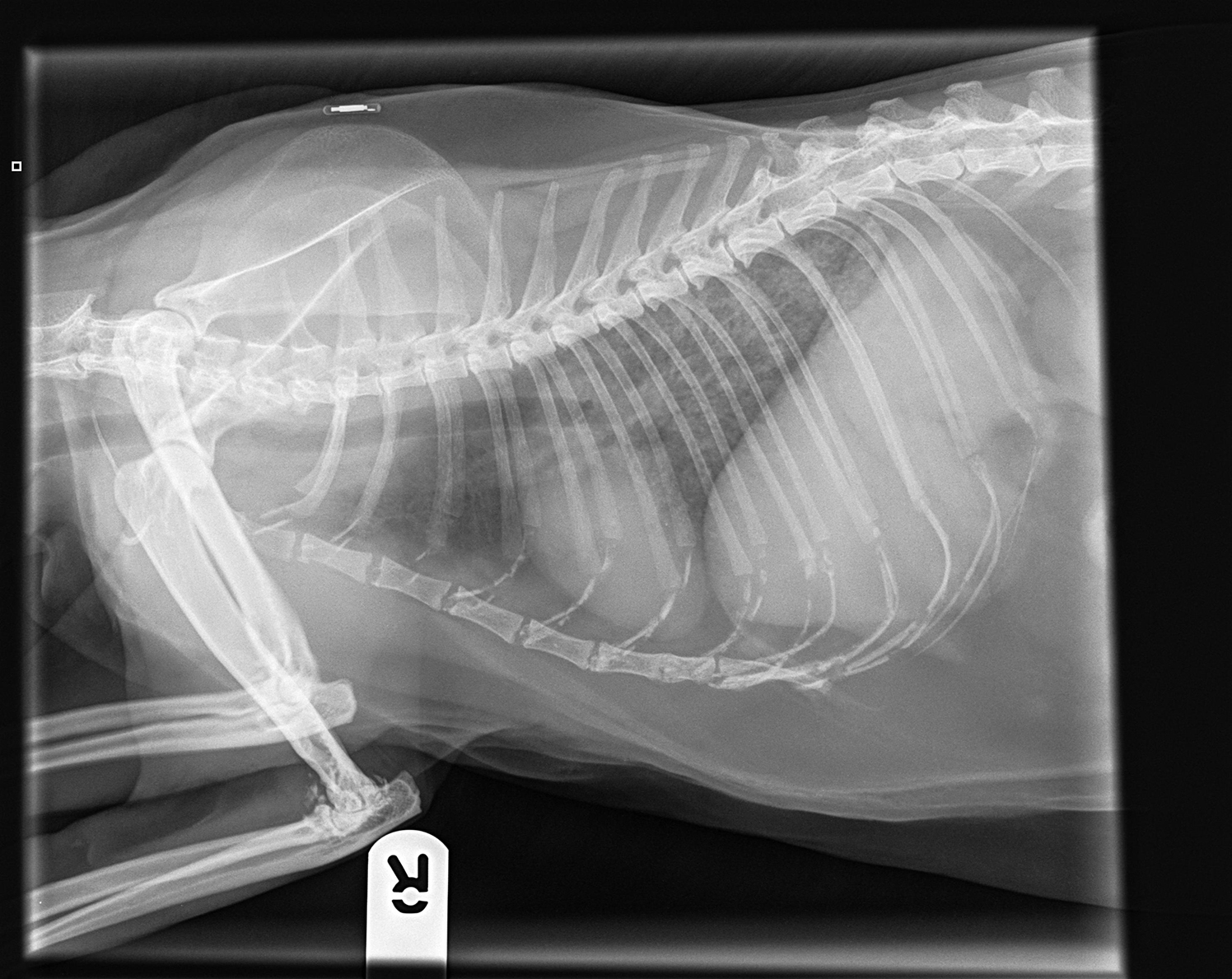
To establish the severity of clinical disease, thoracic radiographs or computed tomography (CT), if available, are required for all cases4,10. This is because many cats develop interstitial lung pathology following initial mycobacterial inoculation and local reaction1-4.
Critically, these patients are very often completely normal on physical examination (thoracic compression and percussion) and auscultation, but can have marked changes as seen on radiographs4.
These changes take longer to resolve than the primary lesion, so monitoring with repeat radiographs is essential and not possible if images from the time of diagnosis are not available.
Due to the potential for adverse drug reactions as outlined later, baseline haematology and biochemistry values are helpful for monitoring purposes4. If possible, ionised calcium should be measured, as hypercalcaemia is a marker of disease severity and possibly a negative prognostic indicator1-4.
Exogenous retroviral testing is also recommended because – although there is no higher prevalence of feline immunodeficiency virus or feline leukaemia virus infections in cats with mycobacterial infections compared to the general population – an infected patient would have a significantly worse prognosis due to their inability to mount an effective immune response1-4,8.
Identifying infecting mycobacterial species
The main tests used to confirm which mycobacterial species is present in a given patient are culture, PCR and interferon gamma release assay (IGRA) tests4,11,12. Specialist mycobacterial culture can only be performed on fresh (not fixed) tissue samples by certain laboratories. In the UK this is APHA Weybridge and is a chargeable service in almost all cases. While this remains the reference test for animal infections, its limitations are the time it can take to culture samples, which can be weeks to months, and its inability to identify fastidious species altogether, for example M lepraemurium4,5. This means the overall sensitivity is approximately 50%1, though it is higher for MTBC species and lower for NTM.
Instead of culture, therefore, PCR testing is now available and widely used. The reference laboratory at The Leeds Teaching Hospitals commercially accepts veterinary samples for testing; however, the sensitivity for non-human samples has not been evaluated.
The PCR can be performed on fresh or fixed tissue, but it is likely that chemicals used in the fixation process can inhibit the PCR reaction and so fresh tissue is preferred where available.
An IGRA test can be used to test cats where there is no tissue available for culture or PCR, as it is performed on blood11,12. The test is available in the UK via Biobest Laboratories, Edinburgh, and has very high sensitivity for MTBC infections, as well as being able to identify some NTM including MAC11.
Treatment
Standard first line therapy for companion animal mycobacterial infections should comprise a fluoroquinolone, a macrolide/azalide and rifampicin, detailed in Table 11-4.
| Table 1. First-line anti-tuberculous medications; a triple combination comprising one of the fluoroquinolones, a macrolide/azalide and rifampicin is frequently used. | ||||
|---|---|---|---|---|
| Drug | Product | Dosing | Contraindications | Side effects |
| Pradofloxacin | Veraflox 25mg/ml suspension or 15mg tablets | 5-7.5mg/kg PO q 24 hours | Not in dogs <12 months of age (18 months in giant breeds) or cats <6 weeks of age due to potential adverse effects on cartilage. Do not use in animals with epilepsy. | Neutropenia (not clinically significant) with high doses and/or long courses. |
| Marbofloxacin | Marbocyl 20mg tablets | 2mg/kg PO q 24 hours | As for pradofloxacin | Intermittent vomiting and transient diarrhoea on instigation of treatment |
| Azithromycin | Zithromax 250mg capsule 50mg/ml suspension | 5-15mg/kg PO q 24 hours | Pre-existing cardiopathy, especially arrhythmogenic disease. Pre-existing hepatopathy. | Intermittent vomiting and diarrhoea possible but uncommon. |
| Clarithromycin | Generic 500mg tablet 50mg/ml suspension | 7.5mg/kg PO q 12 hours | Concurrent use of NSAIDs or antacids. Pre-existing hepatopathy. | Pinnal erythema. Generalised erythema. Hepatotoxicity. |
| Rifampicin | Rifadin 300mg capsule 20mg/ml suspension | 5-10 mg/kg PO q 24 hours by mouth | Pre-existing hepatopathy. Pregnant queens | Hepatotoxicity; induction of liver enzymes, anorexia, generalised erythema and pruritus. CNS a sign. Discolouration of body fluids. |
| PO = per os; CNS = central nervous system; NSAIDs = non-steroidal anti-inflammatory drugs. |
||||
For cats, the fluoroquinolone of choice is pradofloxacin due to its excellent safety profile and its known in vitro activity against mycobacteria, when compared to older fluoroquinolones13. Where this is not available, the use of marbofloxacin can be considered. Enrofloxacin is an unacceptable choice in the cat due to its association with acute irreversible retinopathy; however, it is a good first line agent for dogs.
The azalide of choice for cats is azithromycin due to a dosage pattern of once every 24 hours, which eases adherence to treatment, plus its concentration within pulmonary macrophages4. In dogs, clarithromycin is the preferred choice due to the size of tablets available for accurate dosing.
Rifampicin has long been a cornerstone of anti-tuberculosis, and more general anti-mycobacterial medical therapy2,4. Importantly, it has good efficacy against both extracellular mycobacteria as well as those slowly and persistently replicating within macrophages. However, it should never be used as a monotherapy for mycobacterial disease, as resistance develops rapidly.
Rifampicin is known to be a potent inducer of the hepatic cytochrome P450 system and so has the potential to be significantly hepatotoxic when used chronically. Treatment with rifampicin should be stopped if circulating levels of liver enzymes significantly exceed the upper reference interval, hyperbilirubinaemia develops, or patients develop clinical signs attributable to hepatic dysfunction4. If this occurs, treatment should be stopped and, once the patient has recovered, the rifampicin can be reintroduced at half of the previous dose with close monitoring to ensure the hepatopathy does not recur.
Rifampicin is excreted in all bodily fluids and may cause urine, tears, and saliva to discolour to red-orange in colour. This is not clinically significant, but it can be distressing for clients who are unprepared, especially as the potential exists for the discolouration to be mistaken for haemorrhage. Deposition of the drug within the skin can cause skin discolouration, irritation and, occasionally, hyperaesthesia in both cats and dogs.
Rifampicin is a potential teratogen and so should only be handled by owners wearing gloves and should not be administered to pregnant queens.
Treatment should be given for at least three months and for two months beyond the resolution of clinical signs (including radiographic or ultrasonographic abnormalities), whichever is longer1-4.
This means that, on average, cases with skin and lymph node involvement typically require three months of treatment, whereas those with pulmonary changes as well, or other systemic spread, usually require upwards of six months to ensure resolution. The prognosis was historically guarded, with only 40% of cats reported to respond to treatment2,3.
However, with the implementation of the outlined treatment the prognosis has improved to nearly 80% of cats with MTBC infections causing skin lesions and/or lymphadenomegaly, even with secondary pulmonary spread4. The MAC group of organisms are more resistant to treatment, likely due to inherent drug resistance patterns, so the prognosis is more guarded with only 50-60% of cases achieving clinical remission and typically with longer courses of treatment (usually at least 6 months, but sometimes in excess of 12 months is needed).
Zoonotic Considerations
Before beginning treatment, it is important to ensure that owners and persons in close contact with the patient are fully informed of, and clearly understand the potential zoonotic risks associated with being in contact with an infected animal4,14,15. The greatest risk is posed by members of the MTBC of mycobacteria:
- M tuberculosis. Although rare, infection with M tuberculosis would be considered a significant zoonotic risk. Finding an infected canine should trigger a search for a possible infecting human. Infected animals should be euthanised and their bodies cremated (not buried).
- M bovis. Currently, fewer than 1% of human tuberculosis cases in the UK are caused by M bovis infection. The rare cases of human M bovis TB that have resulted from exposure to cats have occurred where the cats have had skin lesions discharging purulent material containing many organisms. The Health Security Agency consider the risk to humans from M bovis-infected pets to be “very low”15. That said, the risk is still present and should be considered seriously in the context of humans with specific risk factors for transmission (see later) and the clinical signs present4,14,15. Extensive and/or purulent lesions pose the greatest risk to human health and are generally less responsive to treatment4. By comparison, single non-ulcerated skin lesions and/or regional lymphadenopathy carry very low zoonotic risk and may be very amenable to treatment.
- M microti. The risk to humans of M microti is significantly lower than that of M bovis. Very few human cases of infection with this organism have ever been reported, and none have been shown to have resulted from exposure to an infected cat or dog.
- MAC. While not members of the MTBC, this group of organisms can infect humans in the presence of specific risk factors (see later) – again, none have been shown to have resulted from exposure to an infected companion animal.
The following list of risk factors has been compiled from advice published by public health organisations from across the UK and WHO guidelines.
Humans are considered at heightened risk if they:
- are under 5 years old (some sources suggest 12 years)
- are pregnant
- are HIV-positive
- suffer from substance misuse
- have been diagnosed with diabetes mellitus
- suffer (severe) kidney disease
- have ever received a solid organ transplant
- are a cancer patient receiving chemotherapy or radiation therapy
- have any medical condition requiring treatment with systemic corticosteroids
- require specialized treatment for rheumatoid arthritis or Crohn’s disease
- are receiving tumour necrosis factor alpha (TNF-α) inhibitors
In any of the aforementioned situations, owners and veterinarians are strongly discouraged away from animal treatment.
Key Points
- Mycobacterial infections should be included in the list of differential diagnoses for cats with solitary or multiple nodules, ulcers and draining tracts, especially in ‘fight and bite’ locations.
- Acid-fast (for example, Ziehl-Neelsen) staining should always be performed if cytology or histology samples show a granulomatous inflammatory pattern.
- Samples (ideally fresh frozen) should be submitted for culture and/or PCR at specialised laboratories to confirm mycobacterial infection and for species identification.
- The treatment of mycobacterial infections has a good prognosis, but requires long-term therapy with a combination of three antibiotics in most cases for several months.
- Incomplete treatment and lack of compliance are likely to be an important cause of treatment failure and may induce resistance to antibiotics.
- Potential zoonotic risk exists with some mycobacterial species, and this should be discussed with the owner before starting treatment.
Latest news