2 Jun 2014
Health screening of reptiles

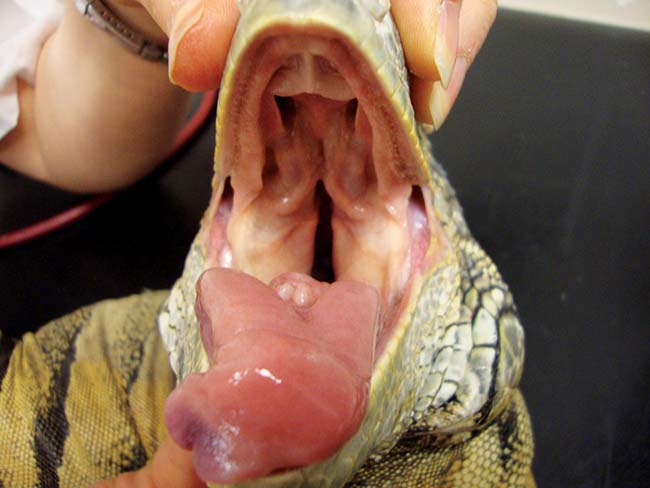
Figure 10. Green iguana with healthy oral tissues.
Health screening is recommended for new animals to be introduced into a collection, as subclinical disease due to infectious pathogens may be present and poses a risk to the individual and the rest of the collection. Common management and infectious concerns are discussed, alongside potential evaluation options.
Newly purchased reptiles may be presented to veterinarians for examination before introduction into a collection, or established collection animals may present for routine annual health checks.
Health screening is used in this article to describe the investigation of the health status of an apparently clinically well animal.
Expectation of the abilities of veterinary professionals by their owners is often high despite a lack of structured training within the veterinary course and often infrequent contact with these animals.
As well as infectious conditions, screening of reptile patients should cover potential management-related concerns, and so the individual animal’s history and the environment, diet and collection management is crucial. Time set aside should be sufficient for taking a detailed history. Owners can fill out details prior to their appointment to speed up the process of history taking without losing accuracy or missing vital details.
To interpret findings, it is very important to be familiar with the requirements of an individual species before evaluation and discussion. Husbandry deficiencies can be factors in the vast majority of clinical problems in pet reptiles. Even if an animal appears well on initial evaluation, if the husbandry is incorrect then subclinical pathology may already be present and health problems will result in the future. A health screen is a good opportunity to evaluate and suggest corrections to the management.
It is recommended time is taken at the beginning of the consultation to discuss husbandry arrangements. While this discussion is taking place there is the opportunity to perform hands-off observation of the animal for a period of time in advance of the complete clinical examination. It is important to make time for this observation and to appreciate respiratory rate, effort and pattern, demeanour, activity and coordination, and response to stimuli. This evaluation is best carried out at the reptile’s normal environmental temperature as extremes of temperature can lead to altered activity and behaviour.
Once history taking and husbandry evaluation is complete then a hands-on clinical examination can be carried out. This can rely upon a checklist or established routine. It is important to be thorough and systematic to avoid any changes being missed. The author starts at the head and evaluates the nostrils, mouth (inside and out), eyes, external ears and facial skin initially, followed by progressive examination and palpation of the body from the nose to tail tip. It is not unusual to find significant health concerns in animals the owner believes to be clinically well as reptiles will generally mask clinical signs until an advanced state of ill health (Figures 1 to 3).



Sexing of the animal should be done in species where this is possible. Some lizards are sexually dimorphic, some require eversion of hemipenes to identify males and others may not be distinguishable by external examination. Snakes may have subtle visible species variation between sexes, but sex is most reliably determined by probing. Typically in male snakes a blunt-ended probe passed caudally from the vent can progress six scales or more into the hemipene sac, but in females a probe passes less than six scales. Chelonia can often be sexed as adults by evaluating physical characteristics such as tail length, vent shape, nail length and plastron shape, but reliability of these characteristics can vary significantly between species so, again, familiarity or a good reference source is invaluable.
Endoparasites
During the examination, many reptiles will pass faeces as a defensive tactic. This provides a valuable opportunity to screen for internal parasites. When screening appointments are booked it is sensible to request owners to bring the most recent faecal sample in case the animal does not oblige during the consult. Every reptile presented should be screened for intestinal parasites. This enables targeted treatment towards specific identified pathogens, but also can give information on the overall health status of the individual. A debilitated or immunocompromised animal may develop high parasite burdens due to an altered host-parasite relationship.
Both direct and flotation concentration techniques can be quickly performed on fresh faeces within practice laboratories.
In anorexic animals, or those in a physiologically normal reduced metabolic state (for example, brumation), faeces may not be passed and a cloacal flush sample can be used instead.
Direct smear
Fresh faeces should be mixed on a slide, with warm isotonic fluid – such as 0.9% saline – and examined immediately. This will demonstrate parasites present in moderate to high concentrations and allow identification of motile protozoa.
A small amount of urates should also be examined to check for renally sourced protozoa such as Hexamita.
Flotation technique
Commercial solutions are available, such as zinc sulphate, or a saturated salt solution can be made easily and used routinely.
Samples should be left in the flotation chamber for 10 minutes to allow parasite concentration, but prolonged periods using hypersaturated solutions can affect parasite morphology and complicate identification.
It is advisable to treat parasite infestations in pet reptiles due to the high risk of escalation of numbers in the captive situation. Retesting is recommended two to four weeks after therapy to ensure individual treatment and environmental decontamination was successful.
Pinworms (oxyurids)
Pinworms are very common in most pet reptile species and can be present in high numbers without detectable adverse effects. The nematode eggs have a characteristic semicircular shape and are often brown in colour (Figure 4).
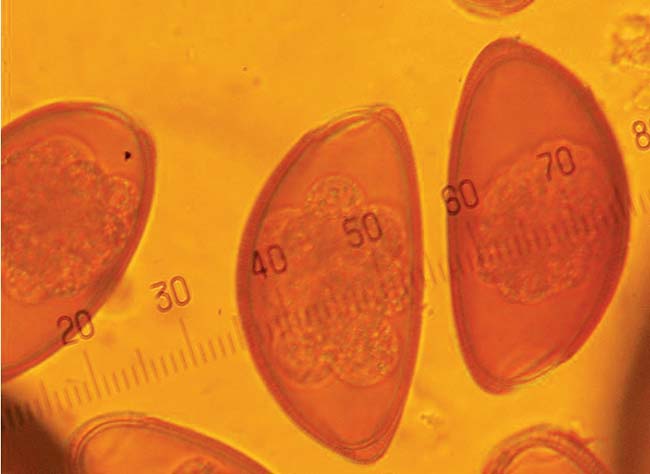
Low numbers of these eggs present in faeces may be considered normal, and good hygiene practice and regular faecal checks should be recommended to ensure numbers do not increase to an abnormal level. Treatment with fenbendazole along with environmental decontamination is recommended where moderate to high infections are noted or clinical signs that may be attributable to parasitic enteritis are present.
Coccidia
Coccidia are most frequently identified in agamids and chameleons (Figure 5). All infections should be treated, and either potentiated sulphonamides or toltrazuril.

Cryptosporidia
Cryptosporidia are protozoal pathogens that infect the gastrointestinal tract. Cryptosporidium saurophilum infection has been reported to cause small intestinal epithelial hyperplasia in geckos and some monitor species, and Cryptosporidium serpentis causes gastric epithelial hyperplasia in a wide variety of snake species. While weight loss in geckos and gastric enlargement and regurgitation in snakes are frequently associated with infection, asymptomatic cryptosporidial carrier states may exist (Figures 6 and 7).


Asymptomatic infections may be detected by examining acid-fast preparations of faecal material, mucus from the surface of regurgitated food and gastric flush fluid, but false negatives are common.
Endoscopic or surgical biopsy of the affected region with histopathological examination to identify the organisms and resulting inflammatory response is recommended.
Potentiated sulphonamides have been found by the author to slow progression of disease in individual animals, but treatment is ultimately unsuccessful and euthanasia should be considered if clinical signs are evident, particularly in the collection situation where infection could be transmitted to other reptiles.
Ectoparasites
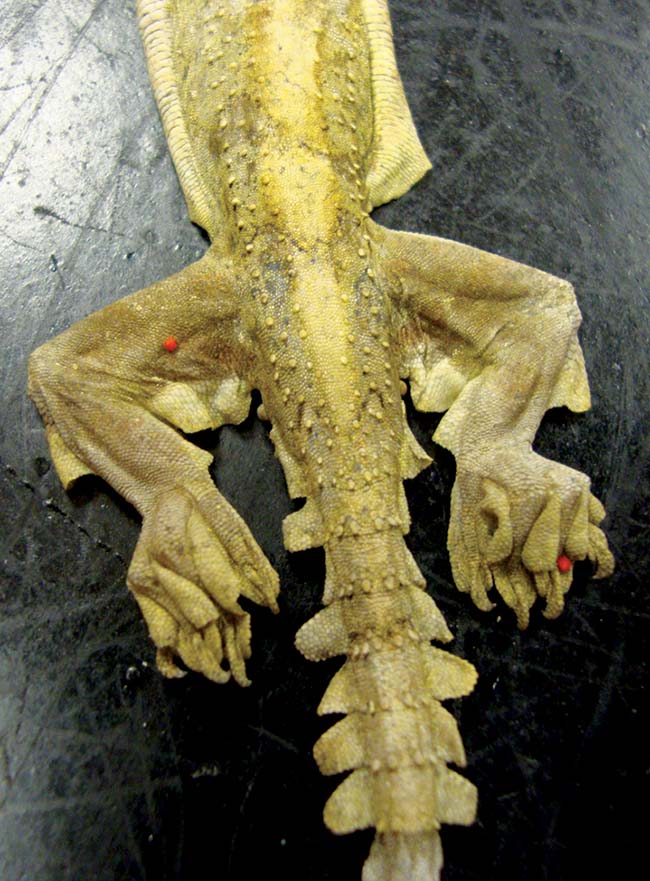
Ophionyssus natricis mites are frequently seen in snakes and lizards. They can be difficult to readily identify as they tend to reside in folds of skin, especially around the eyes. High levels can lead to anaemia or discomfort, but low levels are rarely clinically detrimental. However, treatment is always advised, as these blood-sucking parasites can quickly multiply and may be responsible for transfer of other pathogens.
A variety of other mites may be seen on imported or wild caught animals and should be treated with insecticides, such as ivermectin, fipronil or permethrin (Figures 8 and 9). Any treatments should have the dose calculated based on weight and be carried out in a well-ventilated area to avoid potential overdose and toxicosis.

Non-infectious pathology
Reproductive pathology
Reproductive disease is common in pet reptiles, with lack of social cues, hypocalcaemia, inadequate spectrum or intensity of lighting, inadequate temperatures, lack of oviposition sites and poor health hypothesised as factors. Females with coelomic enlargement or a palpable increase in intracoelomic soft tissue identified on screening examination should be scanned to assess reproductive status.
A more proactive approach is needed to educate owners about husbandry and reproductive disease. Prophylactic neutering of predisposed species has been suggested, but is rarely carried out in pet reptiles. The potential for routine ovariectomy should be discussed with the owners of species predisposed to reproductive pathology, particularly green iguanas and veiled chameleons.
Oral disease
Oral disease may be detected on examination before symptoms are noticeable to owners.
Stomatitis in snakes is typically easily identifiable with hypersalivation, facial swelling, gingival hyperplasia and purulent exudate evident on oral examination. Using a small card to open the snake’s mouth avoids inadvertent injury to snake or vet. The corner of the card is inserted into the diastema at the front of the mouth and manoeuvred across the mouth to prise open the jaws.
Severe disease with advanced infection can be seen with immunosuppression associated with inappropriate husbandry (particularly low or fluctuating temperatures), or underlying viral diseases.
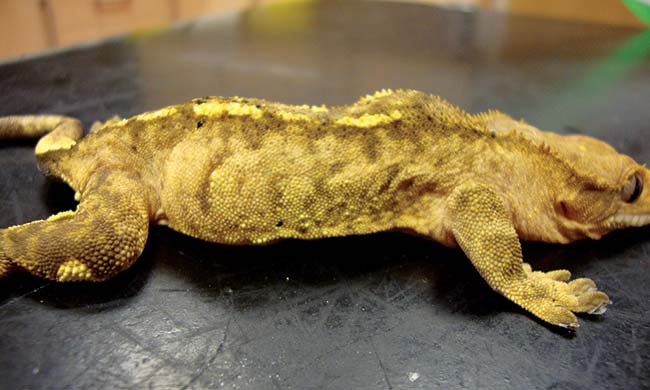
Periodontal disease is common in lizards with acrodont teeth, including dragons, iguanas and chameleons (Figures 10 and 11). These species have teeth that are embedded in the bony jaw and which are not replaced.
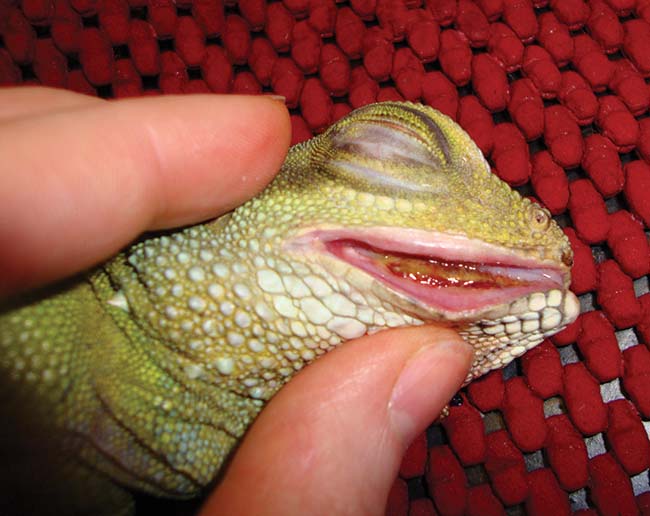
Chronic feeding of an inappropriate soft or high sugar diet predisposes to gingivitis and gingival recession with progressive exposure of the underlying bone. Osteomyelitis and abscessation can result in chronic, untreated cases. Early identification of subtle gingival changes, with correction of diet and removal of plaque or necrotic material, can slow or stop progression of this condition.
Metabolic bone disease
Maintenance of body calcium requires sufficient dietary intake calcium and provision of dietary Vitamin D, with appropriate ultraviolet light provision, heat and normal hepatic and renal function needed to allow endogenous vitamin D formation.
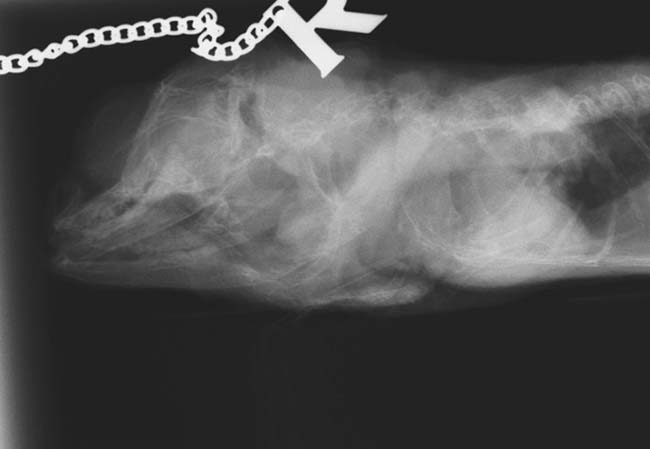
Failure of any part of the homeostatic mechanisms can lead to decreased blood calcium levels. In response, parathyroid hormone is released causing mobilisation of calcium stores from bone. A chronic state of imbalance leads to progressive demineralisation of bone. Pathological fractures, deformed long bones, inability to feed due to pain associated with mandibular distortion, neurological symptoms or chronic wasting can result (Figure 12). This can be prevented if early changes, such as increased mandibular flexibility on compression, or husbandry deficiencies are identified prior to development of advanced changes.
Radiographs to assess bone density, especially of the extremities, can be more informative than a clinical examination in early cases (Figure 13). Screening radiographs are recommended in animals to be used for breeding where husbandry is known to have been inappropriate, or previous husbandry is unknown. Disease is rarely seen in snakes due to their dependence on whole prey items and associated relatively high calcium and vitamin D dietary intake.
Subclinical viral diseases
Ophidian paramyxovirus-1
Ophidian paramyxovirus-1 causes clinical respiratory or neurological disease, but can have a prolonged incubation period of up to a year, so snakes may appear healthy on presentation despite infection.
Serology is possible and paired titres are recommended, especially if snakes are to be introduced into a large or conservationally important collection. A serological response can take up to six weeks to develop so the second titre should be collected at least six weeks after the start of quarantine.
Inclusion body disease
Inclusion body disease primarily causes neurological disease, but can be subclinical (especially in boas). It is believed to be due to viral infection, most probably a recently identified arenavirus. Screening is complicated by inconsistencies in location and number of the histologically typical inclusion bodies. The CNS is recommended for sampling, but is not practical in live snakes. Liver or tonsillar biopsies can be performed for histopathological analysis, but false negatives can occur. Specific testing for arenavirus (PCR and serology) is now available. Mixing of boas and pythons is to be avoided and quarantine periods in snakes should be a minimum of 12 months to minimise risk.
Agamid adenovirus-1
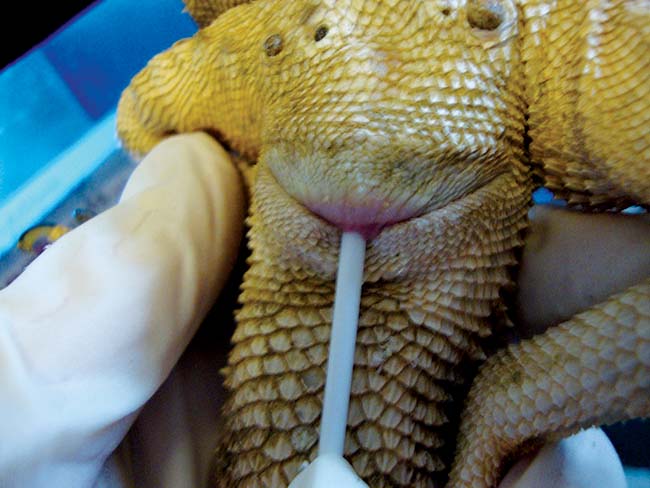
Agamid adenovirus-1 infection is well established in the United States and is believed to be responsible for neurological symptoms, immune suppression, reduced growth and death in infected bearded dragons, but many infected dragons are believed to remain asymptomatic carriers infecting offspring and in-contacts. This adenovirus has also been less frequently isolated from other agamid lizards.
As a large number of bearded dragons have been imported from the US during the past decade to improve breeding stock, the virus is likely to have been introduced and become established in UK populations. Research demonstrates the presence of this virus in clinically normal adult bearded dragons and PCR screening of potential breeding stock is recommended to try to maintain adenovirus free colonies. Sampling is non-invasive and involves swabbing the choana and cloaca to harvest cells for PCR analysis (Figure 14).
Chelonian herpesvirus
Testudo herpesviruses may be associated with necrotic glossitis, stomatitis, upper respiratory infections and, rarely, disseminated infection. In the absence of clinical signs, PCR and cytological techniques lack sensitivity and serology is preferable. Seroconversion can be slow and a quarantine period of six months is recommended by the European Association of Zoos and Aquaria, with serological screening of asymptomatic tortoises at the beginning and end of quarantine. Russian/ Horsfield’s tortoises, spur-thighed tortoises and leopard tortoises appear particularly susceptible to clinical disease and so mixing of species is contraindicated.
Bacterial disease
Mycoplasma
Mycoplasma agassizii is frequently implicated in upper respiratory infections in terrestrial tortoises, but is often carried asymptomatically by clinically well animals. In the absence of clinical signs, PCR techniques lack sensitivity and serology is preferable.
Salmonella
Salmonella (and other Enterobacteriaceae) are endemic among captive reptiles, with prevalence of Salmonella carriage in reptiles reported as up to 94%. Testing and subsequent treatment is controversial, due to the poor success of clearance of infection and potential for development of resistant strains with antibiotic exposure.
Public Health England provides information on preventive measures and is a useful source of information for reptile owners. Routine testing is not recommended due to the insensitivity of testing, high prevalence and lack of treatment options for positive animals. Educating owners about potential zoonoses and good hygiene practice is paramount with pet reptiles being considered as positive and hygiene managed accordingly.
- Please note the medications mentioned in this article are not licensed for use in reptiles and are used via the cascade. Owners should be made aware of this off-licence usage prior to medication administration, along with any potential side effects of treatment.
