25 Jun 2018
Tick-borne diseases in cats
Emi Barker looks disease significance, emerging pathogens, prevention strategies and zoonotic risk regarding the parasite.

Figure 1. An Ixodes species tick attached to a cat. Image: Sophie Tyler.
This article aims to cover recognising which ticks infest cats and the risk factors associated with their presence, appreciating the infectious agents transmitted by the ticks – including those causing non-endemic disease, being able to suggest suitable prevention strategies to concerned clients and being aware of the zoonotic risks of tick-borne diseases.
In a large survey of ticks found infesting UK cats, only those of the Ixodes genus were identified (Davies et al, 2017; Figure 1).
This genus includes Ixodes ricinus (the castor bean or sheep tick), Ixodes canisuga (the dog tick), Ixodes trianguliceps (the vole or shrew tick) and Ixodes hexagonus (the hedgehog tick). In a similar survey of ticks found infesting UK dogs, Ixodes species ticks also predominated (greater than 99% of ticks identified; Abdullah et al, 2016).
The same study also showed Dermacentor reticulatus, the ornate dog tick, to be endemic within the UK – albeit at very low numbers. In contrast, Rhipicephalus sanguineus, the brown dog tick, was only identified on recently imported dogs (Abdullah et al, 2016; Hansford et al, 2017).
The cooler UK climate, compared to the warmer Mediterranean basin, is such that R sanguineus is unlikely to become established in the outdoor environment; however, it is able to infest domestic properties (Hansford et al, 2015). Ticks of genera other than that of Ixodes have been reported to infest cats in continental Europe – these include D reticulatus and R sanguineus (Geurden et al, 2017a).
Risk factors for tick infestation in cats in the UK study were outdoor access, rural environment, middle age (four to six years), and male and un-neutered status (Davies et al, 2017).
These risk factors likely reflect both access to environments where other hosts of these ticks exist and intimate contact with some of these other hosts through predation.
Interestingly, length of coat was not shown to be a risk factor.
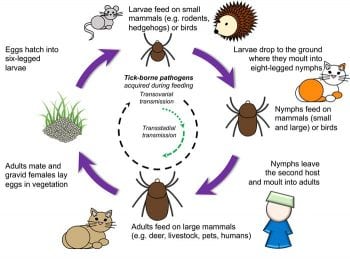
Typically, only nymph and adult forms are found on cats, with small rodents and birds most commonly the host for the larval forms. Attachment and feeding of ticks can cause localised disease, from pruritus through to abscess formation – particularly if incompletely removed. However, their capacity to harbour and transmit a variety of bacterial, protozoal and viral infections – either passively or actively – is the most significant concern.
See Table 1 for a list of tick-borne infectious agents. Many infectious agents are adapted to survive both transstadial and transovarial transmission within the tick (Figure 2).
The public awareness and concern of tick-borne disease has been raised in recent years – in particular regarding the zoonotic agent Borrelia burgdorferi, the aetiological agent of Lyme disease. Outbreaks of infection in untravelled dogs with the previously “exotic” pathogen Babesia canis have also been reported (Woodmansey, 2016a; 2016b; 2017).
Further investigation has demonstrated the presence of B canis within endemic populations of D reticulatus ticks in both south-east England and Wales (de Marco et al, 2017).
What infections are found in ticks from cats?
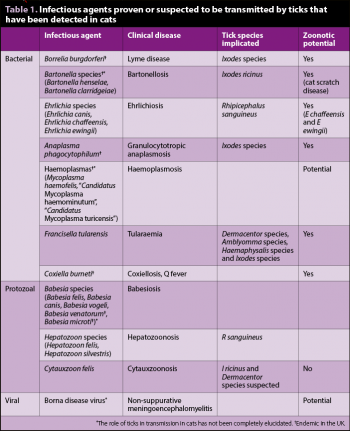
A number of studies have used techniques including serology and PCR to detect evidence of tick-borne infectious agents in both cats and their ectoparasites (that is, ticks and fleas). These studies are not without limitation. Detection of an infectious agent in a cat does not equate to clinical disease in that cat, and for many infections asymptomatic carrier states exist. Also, detection of infectious agents in ticks does not indicate whether that agent originated from the cat from which the tick was collected, or an earlier unknown host.
Analysis of ticks collected from UK cats (Davies et al, 2017; Duplan et al, 2018) revealed low level carriage of many potential pathogens:
- Babesia species (1.1% of samples): isolates closely resembled Babesia microti (0.7%) – the aetiological agent of human babesiosis – and Babesia venatorum (0.4% of samples), which has also been reported to cause human babesiosis.
- Borrelia burgdorferi sensu lato (1.8% samples): isolates comprised Borrelia garinii (1.1%) and Borrelia afzelii (0.7%) – both of which have been associated with human Lyme neuroborreliosis.
- Anaplasma phagocytophilum (0.9% of samples).
- Bartonella species (1.3% of samples): further characterisation was possible in four of the seven isolates; three were Bartonella henselae (0.6%) – a feline pathogen and agent of human “cat scratch disease” – and one was Bartonella clarridgeiae (0.2%) – another feline pathogen and zoonotic agent.
- Haemoplasmas (0.9% of samples): isolates comprised all three of the recognised feline haemoplasmas – “Candidatus Mycoplasma haemominutum“ (0.6%), Mycoplasma haemofelis (n = 1; 0.2%; the agent of feline infectious anaemia) and “Candidatus Mycoplasma turicensis” (0.2%).
- Hepatozoon species (0.4% of samples): isolates comprised one each of Hepatozoon felis (0.2%) and Hepatozoon silvestris (0.2%).
Analysis of ticks collected from cats and dogs in a similar study in Belgium revealed markedly increased carriage of infectious agents in continental Europe (Claerebout et al, 2013). Of the agents tested for, A phagocytophilum, Borrelia species and Rickettsia helvetica (a member of the spotted fever group and suspected human pathogen) were detected in 19.5%, 10.1% and 14.1% of samples, respectively.
This could suggest increased risk of tick-borne disease in cats – and their ticks – even if travel history includes only the northern temperate regions of continental Europe.
Do these ‘tick-borne’ agents cause disease in cats?
The American College of Veterinary Internal Medicine consensus statement on Lyme disease concluded although cats may be infected with Borrelia species, little data support it as a cause of clinical disease (Littman et al, 2018).
Results of experimental infection with the Spirochaetes bacterium B burgdorferi are inconsistent, with some reporting clinicopathological changes that parallel those of human Lyme disease (Gibson et al, 1995) and others that report no changes (Burgess, 1992).
Although Rickettsiales bacteria (Anaplasma species, Ehrlichia species and Rickettsia species) have been detected by PCR in the blood of cats and their ticks, the pathogenic potential of this group is poorly understood in the cat (Pennisi et al, 2017).
Experimental infection with A phagocytophilum – the agent of granulocytotropic anaplasmosis in dogs and humans – in combination with infection with B burgdorferi did not result in clinical signs in one study (Lappin et al, 2015). A small number of UK cats found to be infected with A phagocytophilum in a survey had no associated clinical signs on review of their history (Shaw et al, 2005).
In contrast, a case series of cats naturally infected with A phagocytophilum associated with tick exposure reported lethargy, pyrexia and anorexia (Savidge et al, 2016).
However, clinicopathological changes, where present, were mild-moderate and non-specific, with neutrophil cytoplasmic inclusions identified in only 27% of cases. All affected cats responded to doxycycline (5mg/kg by mouth twice daily).
Facultative intracellular bacteria of the Bartonella genus are more commonly considered a flea-borne disease of the cat than tick-borne, with B henselae detected in 17% of flea samples in the UK (Shaw et al, 2004). Although significant concern exists regarding “cat scratch disease” in humans, and a small number of cases of clinical bartonellosis in cats (typically manifested by cardiac or ocular inflammation), the majority of infected cats appear to be asymptomatic and do not require treatment (Pennisi et al, 2013).

Chronic infection with the epierythrocytic bacteria of the Mycoplasma genus (haemoplasmas) are common, with UK surveys (based on convenience samples) identifying prevalences of 11% to 17% for “Ca M haemominutum”, 1.6% to 2.8% for M haemofelis and 1.7% to 2.3% for “Ca M turicensis”. Although acute M haemofelis infection can induce severe haemolytic anaemia, Ca M haemominutum and “Ca M turicensis” are infrequently associated with clinical disease in immunocompetent cats (Barker and Tasker, 2013).
Tick-borne pathogens also include apicomplexan protozoal parasites of the Babesia, Hepatozoon and Cytauxzoon genera. Babesia felis has been associated with haemolytic anaemia; however, few of the Babesia species found in cats in Europe have been characterised and the significance of their infection remains unknown (Hartmann et al, 2013).
Hepatozoon species infections are rarely associated with clinical disease in cats – likely as the protozoal parasite induces minimal inflammatory response (Lloret et al, 2015b). In Hepatozoon felis infection, parasitic gamonts may be identified at low levels within neutrophils or monocytes on thin blood smear examination, while parasitic meronts most frequently are detected in skeletal and cardiac muscle.
This could account for increases in muscle enzyme activity in some infected cats. In sick cats, concurrent infectious diseases are frequent. Cytauxzoon felis infection is typically rapidly fatal in the US, with signs including severe pyrexia, icterus and haemolytic anaemia; however, the significance of infection with European Cytauxzoon species is less clear as the majority of infected cats were asymptomatic (Lloret et al, 2015a).
As far as the author is aware, tick-borne flaviviruses – including tick-borne encephalitis and ovine encephalomyelitis (louping ill) – have not been reported in the cat, either in association with clinical signs or in PCR-based or serology-based prevalence surveys.
Diagnosis
In cases where tick-borne infection is suspected, serology can be combined with PCR of blood, joint fluid or CSF.
However, interpretation of results should account for duration of disease (for example, in acute disease, seroconversion may not have occurred), likely prior exposure (for example, potential for an incidental positive serology result) and treatment administered (for example, administration of appropriate antibiotics may result in a rapid removal of circulating infectious agents resulting in a negative PCR result).
Repeat (convalescent) serology may be required to confirm clinical suspicion.
Prevention is better than cure
Cats are often subclinical carriers of tick-borne diseases; therefore, control of tick-borne diseases should focus on limiting access to vectors and vector control.
Limiting outdoor access – and, therefore, exposure to environments where ticks reside – appears an obvious solution.
However, as more than 70% of cats have significant outdoor access at one year of age (Rowe et al, 2015), this is not likely a practical solution for many.
Are we being ‘spot on’ with acaricidal treatment?
A survey on feline ticks found no effect of reported administration acaricidal treatment on infestation rate. The authors accepted some owners were unaware the products they were applying were not acaricidal, and most had not reapplied the products at the recommended frequency (Davies et al, 2017). This highlighted the need for owner education.
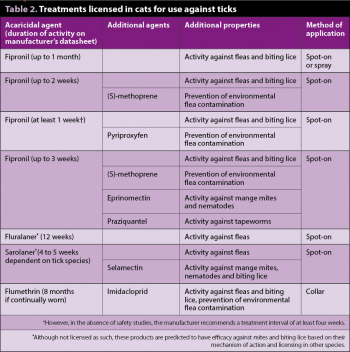
Table 2 shows for a list of products licensed for the treatment of ticks in cats, including suggested duration of efficacy against ticks. Some preparations containing fipronil alone or in combination with (S)-methoprene are available over the counter from pharmacists and other suitably qualified people without a veterinary prescription (legal category: NFA-VPS). Other products are prescription only (legal category: POM-V).
For many years, fipronil was one of the few products with proven acaricidal properties, yet safe for use in cats (Tielemans et al, 2014). However, acaricidal activity is typically shorter than anti-flea activity, and one study suggested efficacy of fipronil varied between target tick species, with efficacy against
I ricinus being less than that against R sanguineus (Geurden et al, 2017a).
Newer products containing isoxazolines (sarolaner; fluralaner) – a novel class of parasiticide – have since become available. Comparative studies, albeit sponsored by pharmaceutical manufacturers, have demonstrated the non-inferiority, if not superiority, of various isoxazolines over fipronil in the management of tick infestation in cats (Geurden et al, 2017a; 2017b).
Both fipronil and isoxazolines are selective inhibitors of gamma-aminobutyric acid-gated chloride channels in arthropods and L-glutamate-gated chloride channels. They result in paralysis in both fleas and ticks. Clients should be warned, as attachment and feeding of individual ticks cannot be completely prevented by these measures, regular owner inspection of the cat’s coat and immediate removal of attached ticks using appropriate tools (Figure 3) is still required.
It should be noted neither fipronil nor the isoxazolines have tick-repellent properties and are reliant on feeding for efficacy. In contrast, a collar containing slow release imidacloprid/flumethrin has been shown to have both acaricidal and repellent properties (Fourie et al, 2015).
The acaricidal and repellent properties are due to the synthetic pyrethroid flumethrin, which inhibits arthropod sodium channels resulting in paralysis. However, it has the disadvantage of being a continually attached collar, which some owners may be reluctant to apply.
Zoonotic implications
All ticks found to infest both cats and dogs have the potential to parasitise humans – either as a result of shared exposure to the outdoor environments where the ticks reside, or the pet bringing the tick into the home environment.
Cats may act as a reservoir for zoonotic infection or merely as a mechanical vector for a tick infected with a human pathogen. Potentially of greater concern is cats with a history of tick exposure – particularly those with a history of travel – would also have been exposed to other arthropods and their infectious diseases (for example, sandflies and Leishmania infantum; mosquitoes; and West Nile virus). Fortuitously, preventing or minimising tick exposure would likely be effective at reducing the exposure to other arthropods and their diseases.
The risk to humans of acquiring tick-borne infection from their cats is very low (Day, 2016). Dogs are considered a reservoir for a greater number of tick-borne infectious agents and the prevalence of tick infestation in cats is lower than that of dogs (Abdullah et al, 2016; Davies et al, 2017). The zoonotic potential ofsome tick-borne diseases (for example, Borna disease virus) has yet to be fully explained (Lutz et al, 2015) and very little data are available to show cats could pose significant reservoirs for human disease.
Conclusions
Tick-borne infections are uncommon in cats, and their role in feline disease is poorly understood. Prevalence is greater in continental Europe, such that tick-borne disease should be considered more likely in cats with a history of travel.
Minimisation of risk of tick-borne infection is multimodal and, where acaricidal products are administered, close attention to treatment intervals is required to maintain coverage.
Cats represent a low risk to owners as vectors of zoonotic tick-borne diseases, but could be considered as sentinels to exposure of tick-borne, or other arthropod-borne, disease.
Latest news

Small animal
Expert Insights: The role of monoclonal antibodies (mAbs) in osteoarthritis (OA) management for pets
Sponsored
10 Mar 2025